
Helium Shortage: Hydrogen as an Alternative Carrier Gas with Benefits on Top
The use of helium as a carrier gas in gas chromatography (GC) has been the go-to option for many years. It has been a popular choice due to its inertness, excellent separation performance, low safety risk, and fast analysis times. However, the worldwide reserves of helium are limited, and this has led to a shortage, resulting in delivery issues and high prices. Therefore, laboratories might be facing severe downtimes and, in some cases, even insolvency. Despite these disadvantages, it remains the preferred carrier gas. This article will demonstrate that hydrogen as an alternative to helium even comes with advantages like high separation efficiency and reduced analysis time up to 40%.
Alternatives to Helium: Nitrogen and Hydrogen
Regarding the three most commonly used carrier gases in GC, the Van-Deemter plot can provide valuable insights. This plot illustrates the height equivalent of a theoretical plate (HETP) in relation to the linear velocity of the gas. A smaller HETP value indicates a higher separation efficiency of the system.
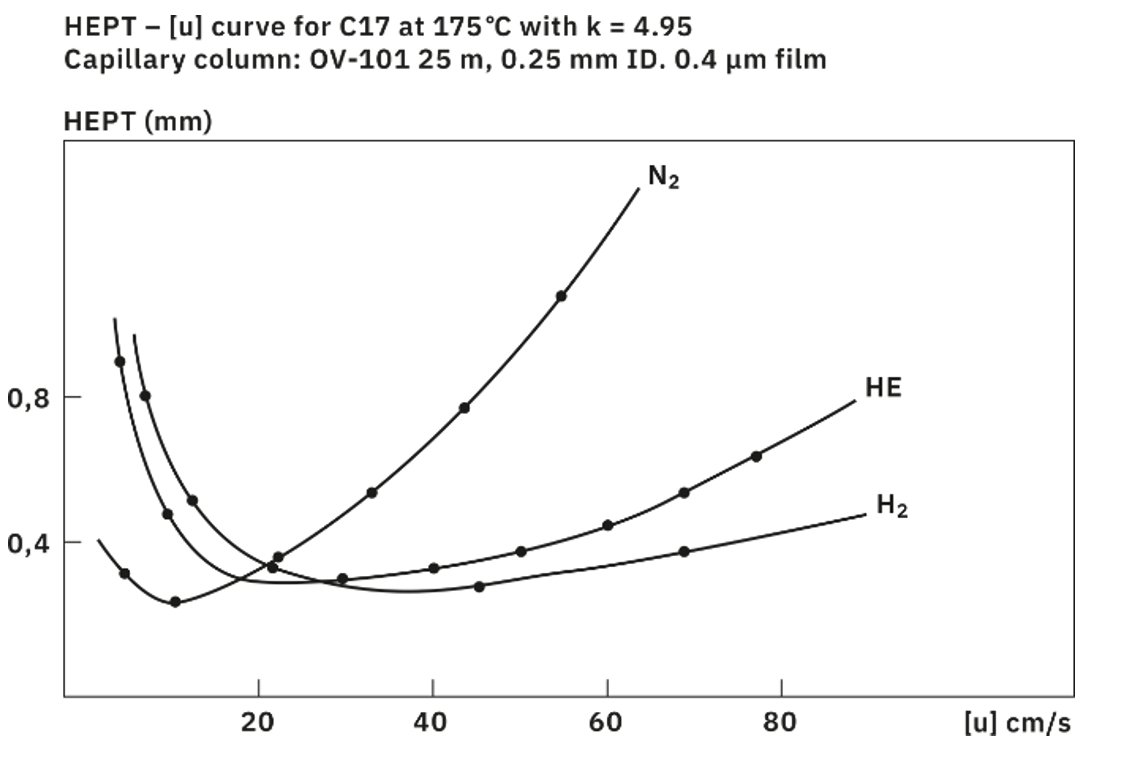
Nitrogen exhibits the lowest HETP. However, its separation efficiency significantly declines at higher or lower flow velocities than the optimum. Nitrogen requires a longer time to achieve satisfactory separation results. Moreover, its low diffusion coefficient limits its applicability for time efficient routine analysis.
In contrast, helium has a slightly higher minimal HETP and attains optimum separation efficiency at a higher flow rate compared to nitrogen. Together with its inertness, this characteristic is often why helium is preferred in laboratory and routine settings.
Hydrogen, on the other hand, is comparable in terms of HETP with helium, but its unique properties make it an attractive alternative to helium. With its low viscosity and high diffusion coefficient, hydrogen can achieve much higher linear velocities maintaining excellent separation performance. Moreover, another advantage of hydrogen lies in its availability and the possibility of on-site generation using gas generators, reducing dependence on external suppliers.
However, the use of hydrogen does present challenges. Firstly, hydrogen is highly reactive and flammable, demanding specialized safety precautions in the laboratory. Secondly, the combination of hydrogen with GC-MS (mass spectrometer) can be problematic, as older GC-MS pumps may not be capable of handling the high flow rates required for hydrogen. Although new GC-MS system can manage these hydrogen flow rates, there is a significant loss of sensitivity (typically a factor of 3–10) when using hydrogen as a carrier gas in GC-MS.
Benefits by Using Hydrogen
The choice of carrier gas in gas chromatography depends on the specific application and analysis requirements. However, as mentioned earlier, hydrogen offers numerous advantages, including more than 40% reduced analysis time. Furthermore, there are straightforward solutions to address the challenges mentioned.
The risk of achieving an explosive hydrogen concentration in a laboratory is almost non-existent due to standard venting systems. Additionally, GC vendors such as Shimadzu provide multiple safety features in their instruments, such as a leakage detection sensor inside the GC that automatically shuts down hydrogen supply in the event of an anomaly. When combined with a hydrogen generator, the risk of reaching an explosive concentration of hydrogen in the GC or laboratory is essentially eliminated.
The biggest drawback of hydrogen, a generally reduced sensitivity in classical GC-MS systems, can be overcome by switching from GC-MS to LC-MS instruments. The pumps in LC-MS systems are powerful enough to handle even much higher flow rates of hydrogen enabling a further speedup of the analysis. The unique plug-and-play SICRIT® Ion Source enables the coupling of a GC with an LC-MS, harnessing the advantages of both techniques for your measurements. Furthermore, when hydrogen is used in combination with the SICRIT® Ion Source and an LC-MS system, there is no loss, but even a gain in sensitivity.
Our recent App Note „GC-SICRIT®-MS: Fast and sensitive analysis of 6 nitrosamines by replacing helium with hydrogen as carrier gas“ shows that hydrogen can be used as a carrier gas without a loss of sensitivity and additionally can speed up your analysis time by almost 50%.
Rethink your Lab
Laboratories will need to find alternatives to helium as its worldwide reserves are limited, leading to delivery issues and high prices. Additionally, in the interest of sustainability, it is crucial to rethink laboratory workflows and explore suitable alternatives in GC. This is especially important as the remaining supply of helium is also essential for medical applications like MRI where no viable substitutes have been found thus far.
Are you ready to rethink your lab? Contact us for more information!
