AI-generated illustration (created with ChatGPT image generation)
The Molecular Ion Problem in GC-MS Pesticide Analysis: A Known Limitation That Still Matters
Gas chromatography–mass spectrometry (GC-MS) is a well-established tool in pesticide residue analysis. Its robustness and reproducibility have made electron ionization (EI) the standard ionization method for decades.
However, EI comes with an inherent limitation. It produces extensive fragmentation, and in many cases, the molecular ion is weak or not visible at all. This directly affects how confidently compounds can be identified.
What is the molecular ion problem
In electron ionization, molecules are exposed to high-energy electrons, leading to fragmentation into multiple smaller ions. While these fragment patterns can be characteristic, the intact molecular ion is often missing or too weak to be reliably used.
This creates a fundamental limitation. Without a clear molecular ion, the exact mass of the compound is not directly observable. Identification relies heavily on matching fragment patterns rather than confirming the intact molecule.
In practice, this can lead to ambiguity, especially when dealing with structurally similar compounds or complex mixtures.
Why it matters in pesticide analysis
Pesticide analysis adds additional layers of complexity. Samples often contain co-eluting compounds from food or environmental matrices, and analytes are frequently present at low concentration levels.
Under these conditions, fragment-based identification becomes more challenging. Matrix can overlap with diagnostic fragments, and small differences between compounds may be difficult to resolve.
The absence of a reliable molecular ion becomes a bottleneck. It limits confidence in identification, particularly when results need to support regulatory decisions or scientific conclusions.
Implications for MS/MS and confirmation workflows
Modern mass spectrometry increasingly relies on MS/MS for structural confirmation. These workflows depend on the selection of a well-defined precursor ion.
In conventional GC-EI workflows, this presents a challenge. EI spectra acquired at the MS¹ level are often not readily compatible with MS/MS approaches, as the molecular ion is frequently weak or absent. Without a clear precursor m/z, it becomes difficult to implement MS/MS acquisition strategies or to integrate these datasets into MS/MS-based pipelines.
As a result, laboratories working with GC-EI often face limitations when trying to adopt newer MS/MS workflows. Existing spectral libraries are largely based on fragment patterns rather than precursor-driven fragmentation, making it difficult to translate legacy data into MS/MS-compatible formats.
This discrepancy between fragment-based EI data and precursor-driven MS/MS workflows limits the ability to combine both approaches within a unified analytical strategy.
A different perspective with molecular ion focused ionization
An alternative approach is to shift the ionization process away from fragmentation and toward the preservation of the molecular ion.
GC-SICRIT® introduces soft ionization, applied downstream of the GC separation. Instead of extensive fragmentation, this approach promotes the formation of molecular and quasi-molecular ions such as [M+H]+ or [M]+.
In qualitative studies covering more than 160 GC-amenable pesticides, this ionization behavior was consistently observed across compound classes. Compared to EI spectra, the resulting data are dominated by intact molecular species with reduced fragmentation.
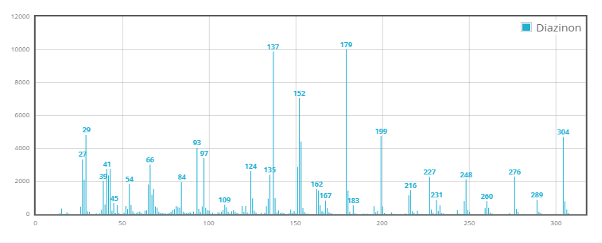
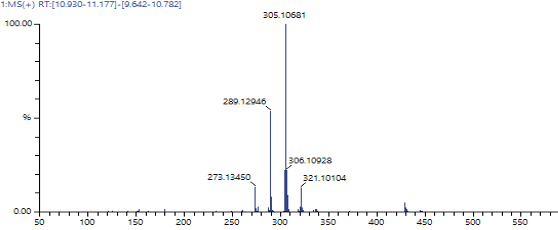
A representative example is Diazinon. While EI produces a highly fragmented spectrum, GC-SICRIT® yields a dominant molecular ion, enabling direct precursor selection for MS/MS and more straightforward interpretation.
Diazinon Spectrum ionized via GC-EI
Diazinon Spectrum ionized via GC-SICRIT®
What changes in practice
The presence of a stable molecular ion changes how GC-MS data can be used.
A clearly defined precursor enables reliable MS/MS acquisition, improving structural confirmation. Spectra become easier to interpret, as fewer fragments compete for attention and matrix overlap is reduced.
In addition, molecular-ion-based spectra are more compatible with existing MS/MS libraries. This opens the possibility to use established LC-MS/MS databases for compound confirmation in GC-based workflows.
The overall result is a shift from fragment-driven identification toward more direct, structure-oriented confirmation.
Additional practical advantages
Beyond spectral quality, molecular ion focused ionization also affects workflow efficiency.
In many GC-MS methods, derivatization steps such as trimethylsilyl (TMS) derivatization are used to improve volatility or ionization efficiency. With softer ionization conditions, direct analysis becomes feasible for a broader range of compounds, potentially reducing or eliminating the need for derivatization.
This simplifies sample preparation, reduces potential sources of error, and shortens overall analysis time.
Another practical aspect is gas usage. Conventional GC-MS often relies on helium both as a carrier gas and within the ionization process. In GC-SICRIT® configurations, ionization is independent of helium, allowing the use of alternative carrier gases such as hydrogen.
This reduces dependency on helium, lowers operational costs, and supports more sustainable laboratory operation.
What this means for laboratories
For research and analytical laboratories, these changes translate into tangible benefits.
Molecular ion visibility improves confidence in compound identification. MS/MS workflows become more accessible and reliable. Data interpretation is simplified, particularly in complex matrices.
At the same time, workflow efficiency can be improved through reduced sample preparation and more flexible gas usage.
Taken together, this represents a meaningful extension of conventional GC-MS capabilities rather than a replacement.
Conclusion
The molecular ion problem in GC-MS is well known, but its impact on modern analytical workflows is often underestimated.
As pesticide analysis moves toward higher confidence, MS/MS-based confirmation, and broader compound coverage, the availability of a reliable molecular ion becomes increasingly important.
Molecular ion focused ionization offers a different analytical perspective, enabling GC-MS data to be used in new ways while maintaining the strengths of gas chromatography.
This post was created with the assistance of AI and editorially reviewed.